For best results, mix ingredients at room temperature. Prep and baking time: 20 minutes. Chill time: 1 hour.
Ingredients
- 125 g butter
- 1 egg
- 125 g sugar
- 1 pinch of salt
- Aroma: zest of half a lemon or lime, or 2-3 drops of lemon or vanilla or almond extract
- 250 g flour
Notes
Any cookie recipe works great as long as you avoid using raising agents in your mixtures and stick to plain flour over self-raising flour.
Try science!
Instructions
- Stir softened butter until it forms peaks.
- Add the egg, sugar and salt and mix until mixture is smooth.
- Add the aroma and flour and mix well.
- Cover the dough and chill in the fridge for 1 hour.
- Roll out your dough to 4-7 mm thickness and cut out your design, transfer to the baking paper, and apply the stamp (with the cutter still around) there. Use a small amount of flour to dust over the dough surface, cutter and rolling pin. Use plenty of flour underneath the dough to facilitate transfer to the baking paper.
- For best results further chill for half an hour before baking.
- Bake at 170-200 °C for 10-15 minutes. Leave to chill before decorating and serving.
- Clean your science cookie cutters in warm water with plenty of soap, preferably with a brush. Our cutters are NOT dishwasher safe and will warp at temperatures of around 60°C.
Science Sugar Cookies Recipe for Chemists
Ingredients
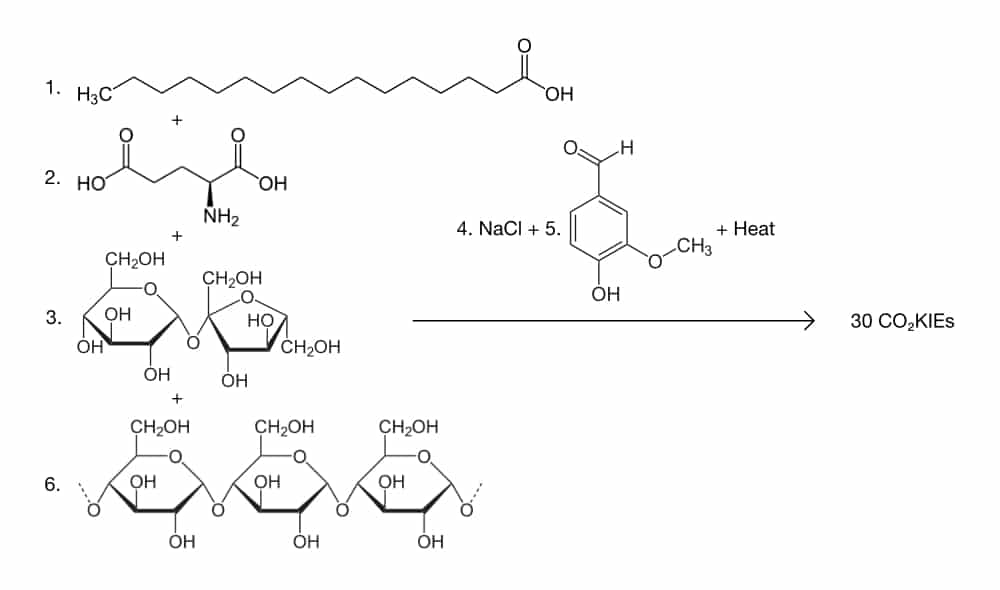
- 125 g partially hydrogenated tallow triglyceride
- one (60 g) calcium carbonate – encapsulated avian albumen + vitellus – coated protein (glutamic acid, aspartic acid, leucine, etc.)
- 125 g sucrose (C12H22O11)
- 0.3 g sodium chloride (NaCl)
- 0.6 g 4-Hydroxy-3-methoxybenzaldehyde (C8H8O3)
- 250 g amylum, water, gluten mixture (73:15:12)
Instructions
In a 2-L round reactor vessel with an overall heat-transfer coefficient of about 570 W/(m²·K) (100 Btu/F-ft2-hr) and a radial flow impeller operating at 100 rpm preheat one to 293.15 K and stir until it forms peaks.
Add ingredients two, three, and four until the mixture is homogeneous. Now add five slowly and with constant agitation. Care must be taken at this point in the reaction to control any temperature rise that may be the result of an exothermic reaction.
When no further temperature rise is observed let the mixture sit without stirring and cool to about 283.15 K for one hour.
Using a screw extrude attached to a #4 nodulizer place the mixture piece-meal on a 316SS sheet (300 x 600 mm). Transfer 316SS sheet to a glove box and flatten the pieces to a thickness of 5 mm before cutting and stamping them with the appropriate science cookie cutters.
Heat the resulting forms in a 463.15 K oven for a period of time that is in agreement with Frank & Johnston’s first order rate expression (see JACOS, 21, 55), or until golden brown (approx. 0.16 hour)
Once the reaction is complete, place the sheet on a 298.15 K heat-transfer table allowing the product to come to equilibrium.
The theoretical yield is 30 CO2KIEs.
Inspiration: Chemical & Engineering News (C&EN, Jun 19, 1995, p. 100). Jeannene Ackerman of Witco Corp.
